
In this notation, the anode, or oxidation half-cell is on the left and the cathode, or reduction half-cell is on the right. A convenient short-hand notation for this is: We can understand the direction of electron flow by breaking this into half-reactions:Įlectrons are leaving the Zn half-cell and going towards the Pb half-cell. The schematic shows the cell set up for the spontaneous reaction between Zn metal and Pb 2+ ions: The images to the right show what a carrou cell looks like in the laboratory, and how we depict it schematically. The paper is wet with the salt solution, to ensure ions can flow through the paper. 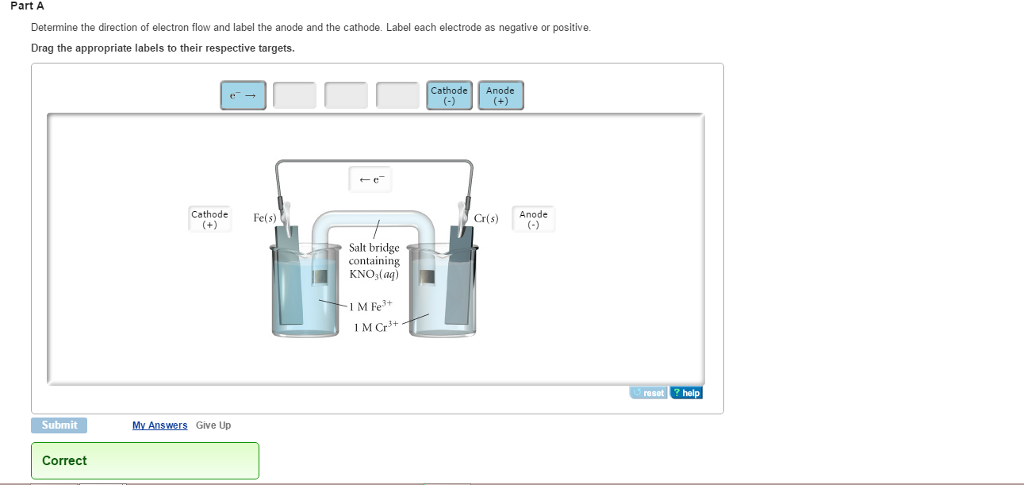
This is done by filling the center well with a salt solution, and using paper to connect each of the outer wells to the center well. The salt bridge connecting two of these outer wells passes Each of the outer wells holds a half-cell. Number of wells that serve as the above beakers. In our laboratory, we will be using a Carrou cell, which provides a more convenient means to carry out experiments on electrochemical cells. The flow of electricity in the circuit is maintained by the electrons moving through the wires and the ions moving through solution. The electrons travel between the electrodes on the wire and never enter the solution. Keep in mind that in an electrochemical cell, only the ions travel in solution. Video explains this setup and details the process at a molecular level:Īfter watching the video, please complete the diagram below to identify the key components in an electrochemical cell: We then connect these cells together (using a wire and a salt bridge) to create an electrical circuit. For the reduction half-cell, a strip of Cu metal is placed in a The oxidation reaction, a strip of Zn metal is placed in a solution of Zn 2+ ions. The electrochemical cell consists of two "half-cells" that correspond to each of the above half-cell reactions. The electrochemical cell forces the electrons to flow through a wire as they go from Zn to the Cu 2+ ions. Previously, we wrote half-reactions that illustrate the nature of this electron flow: 
In this redox reaction, electrons are transferred from Zn to Cu 2+. In the movie on the previous page, when a zinc strip is placed in a solution with Cu 2+ ions, the following spontaneous redox reaction occurs: Electrochemistry: Galvanic Cells and the Nernst EquationĮlectrochemistry Tutorial: Galvanic Cells and the Nernst Equation > Step 2: The Electrochemical CellĮlectrochemistry: Galvanic Cells and the Nernst Equation Step 2: The Electrochemical Cell The double vertical lines refer to the salt bridge - note that the salt bridge must be an inert salt to both ions present in both compartments of the galvanic cell. The single vertical lines indicate the boundary (phase difference) between solid #Ni# and #Ni^(2+)# ions in the aqueous solution of the first compartment and between solid #Ag# and #Ag^(+)# ions present in the aqueous solution of the second compartments. Start with the anode components (site of oxidation) - the cathode components are listed to the right. 
Note that the anode is the site of oxidation (where electrons are lost) and the cathode (where electron are gained) is the site for reduction. Manipulate the two equations so that #E_(cell)^o # is positive. In the galvanic cell, the reaction is spontaneous and for a spontaneous reaction #E_(cell)^o # must be a positive quantity. Normally, the values are listed at the back of most chemistry textbooks. Start by finding the standard reduction potential for the #Ag^+ # and #Ni^(2+)# ion.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
